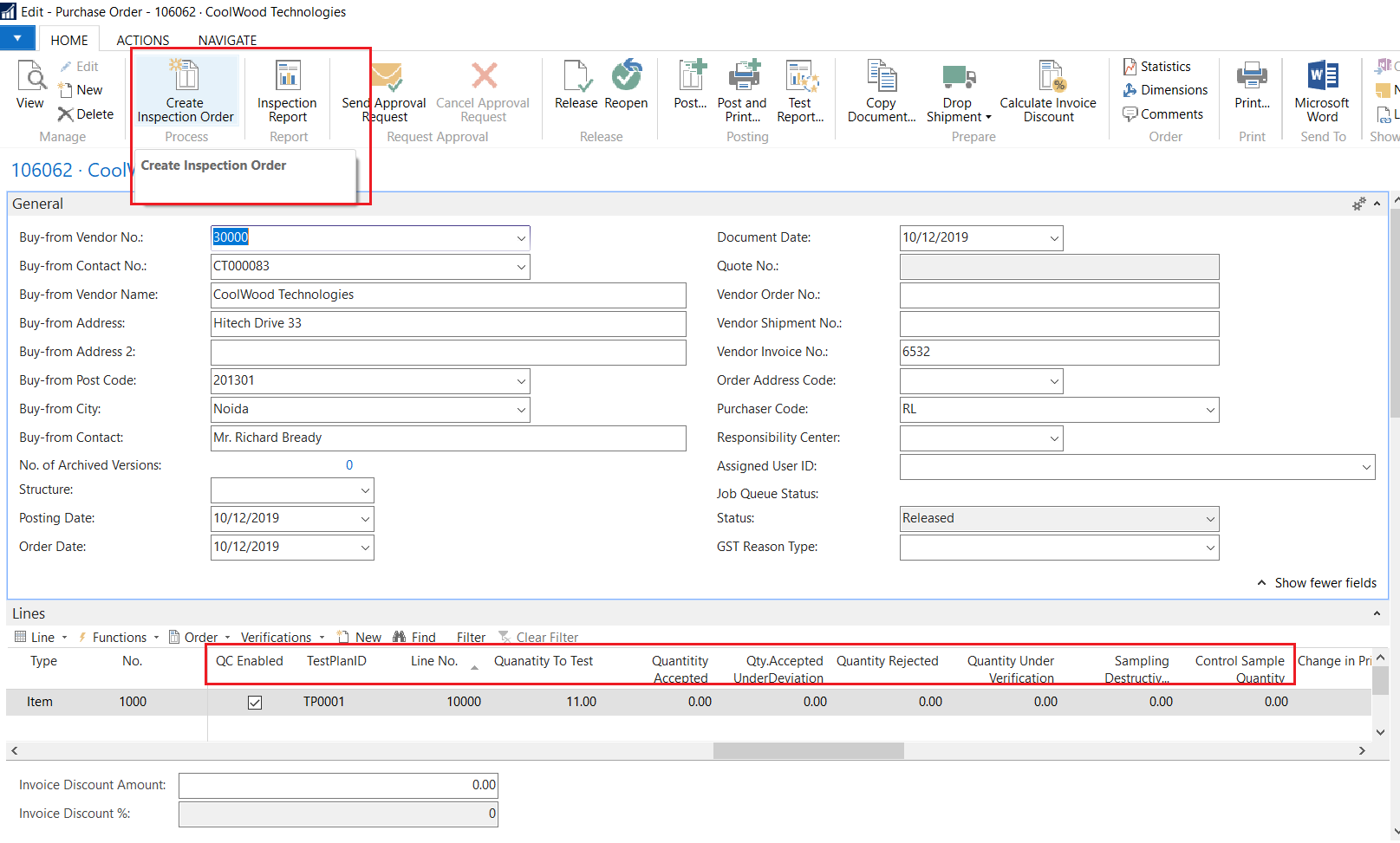
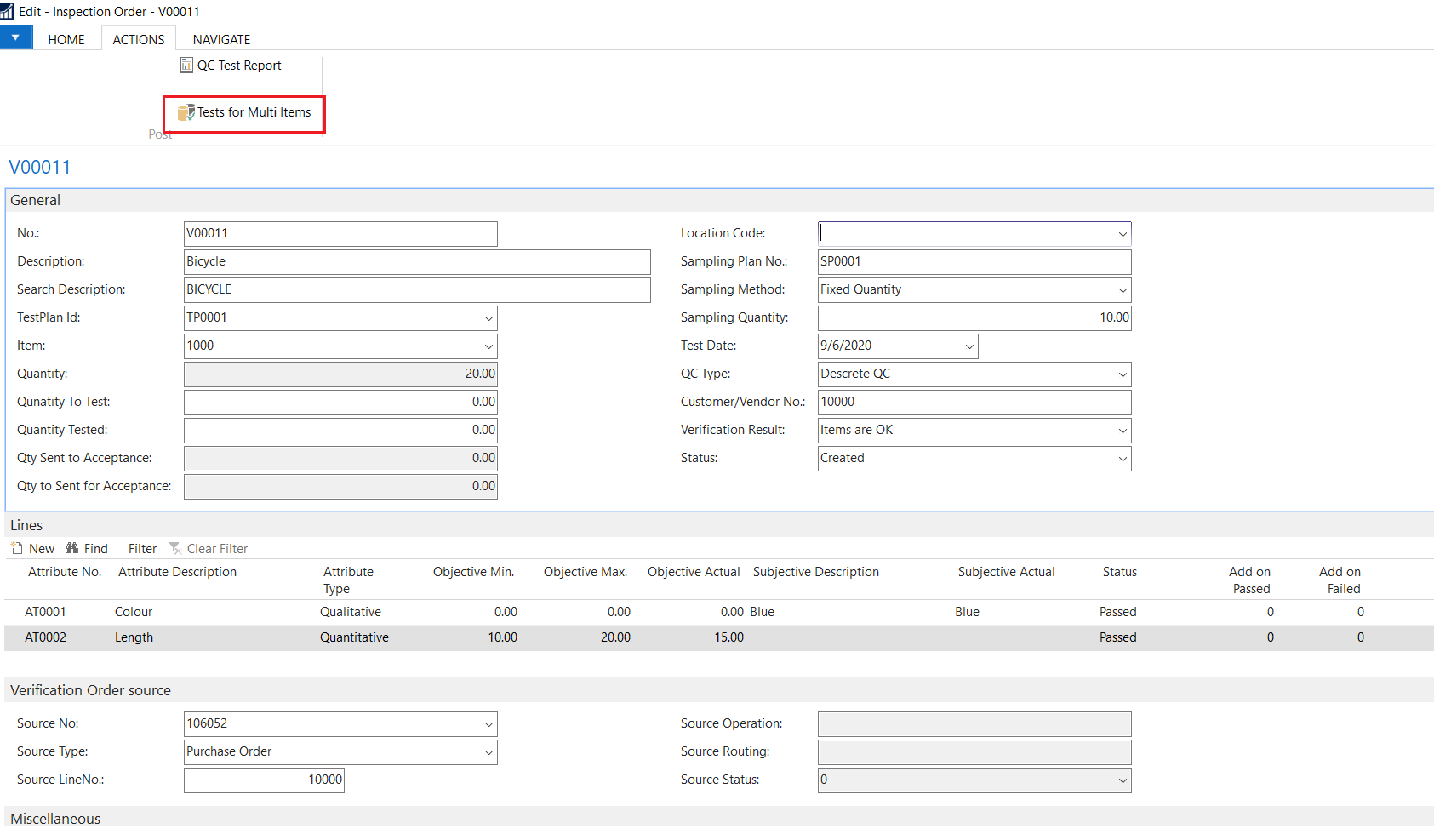
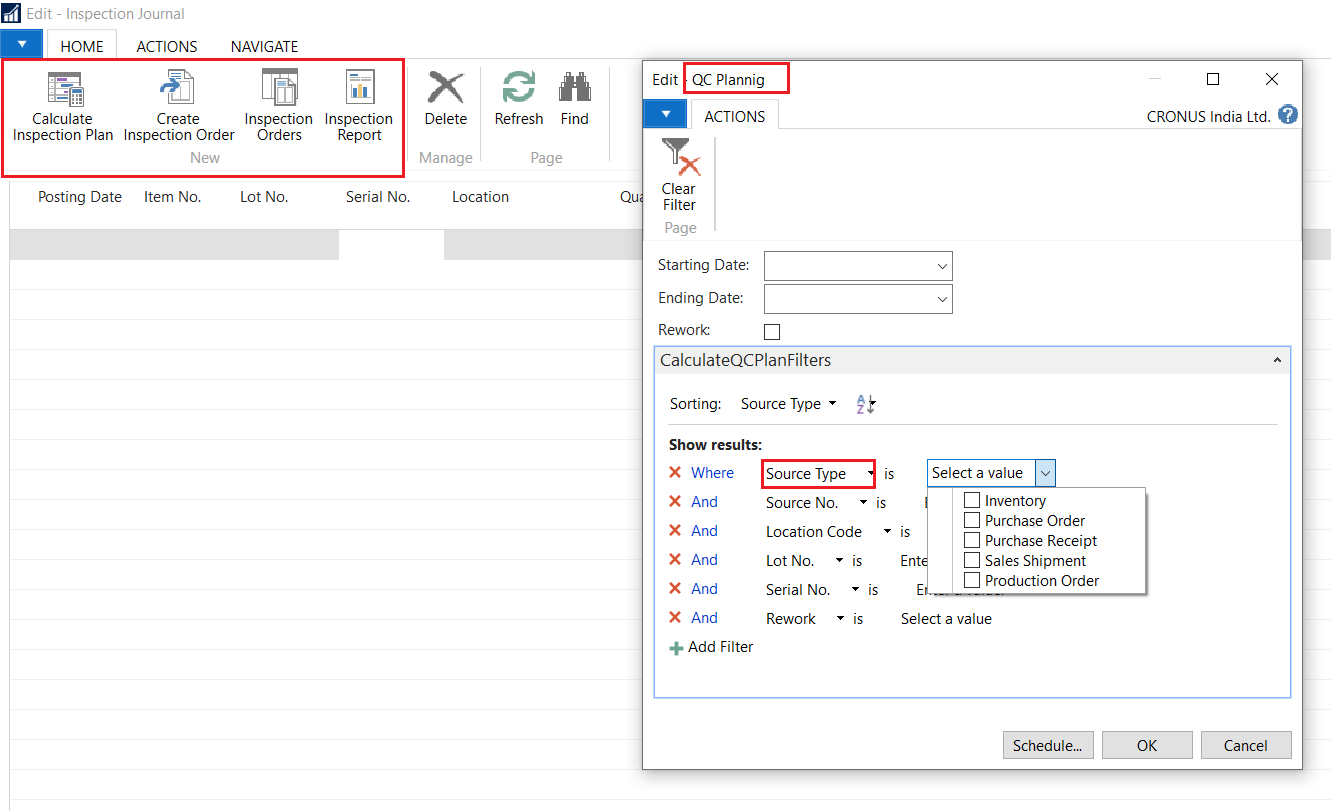
Quality control (QC) is a process through which a business seeks to ensure that product quality is maintained or improved. Quality control requires the business to create an environment in which both management and employees strive for perfection.
- Quality control is a process that is used to ensure a certain level of quality in a product or service.
- It might include whatever actions a business deems necessary to provide for the control and verification of certain characteristics of a product or service.
- Most often, it involves thoroughly examining and testing the quality of products or the results of services.
- The basic goal of this process is to ensure that the products or services that are provided meet specific requirements and characteristics, such as being dependable, satisfactory, safe and fiscally sound.